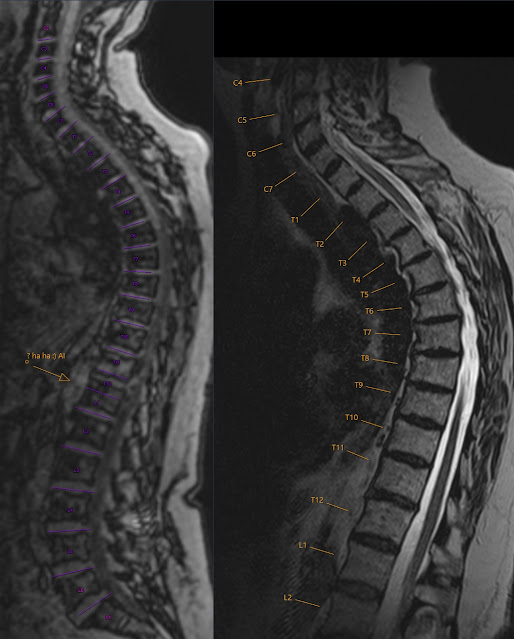
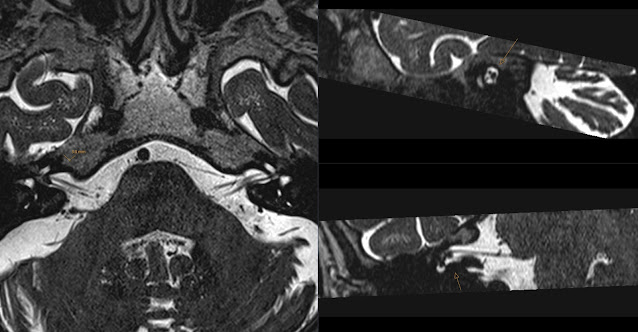
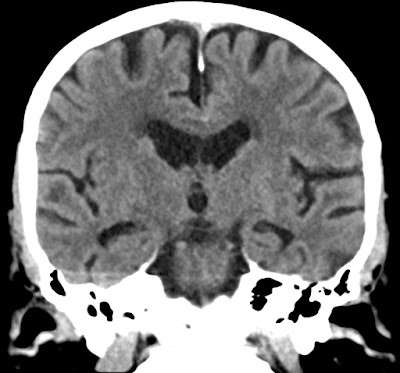
X-mas presents from AI

I asked ChatGPT-4: "Can you make me an image of a simulated Lego set of an MRI Scanner. Show it in foreground with its box behind." This is what I got: This image was generated by AI called Dall-E that is subdivision of ChatGPT. It looks pretty impressive! It would certainly be a great present for X-mas! I was inspired to make this prompt after seeing the image below that i found on X, that also looks like generated by AI. I think capabilities of AI in the field of Radiology are pretty impressive! But honestly I was even more impressed when I asked ChatGPT to spot the difference between two images I took in my garden. It did the great job and this is quite promising example of how can AI be used in Radiology. See the images for yourself first and spot the difference: And now look at result of AI analysis: As a fun task I asked AI to make me an image of a cat sitting on top of aquarium - something I'm used to see in my office. Here is what I got: I think it's very